Advantages
- Superior Microenvironment Modulation: Successfully induces a phenotypic shift from pro-tumorigenic M2 macrophages to anti-tumorigenic M1 macrophages, significantly promoting T-cell infiltration.
- High Synergy with Immune Checkpoint Inhibitors (ICIs): In combination with anti-PD-1 antibodies, it demonstrates remarkable synergistic tumor suppression that cannot be achieved by monotherapy.
Background and Technology
In the field of cancer immunotherapy, a key emerging strategy is the reprogramming of tumor-associated macrophages (TAMs) from the immunosuppressive M2 phenotype to the activated M1 phenotype, which releases nitric oxide (NO) and possesses anti-tumor activity. L-arginine serves as a critical substrate: in M1 macrophages, it is metabolized by the enzyme iNOS to produce NO, whereas in M2 macrophages, it is metabolized by arginase into substances that promote tumor growth. Furthermore, NO exhibits "biphasic" properties, where low concentrations promote angiogenesis and tumor progression, while high concentrations induce cancer cell death. Consequently, effective therapy requires precise delivery to favor iNOS-mediated metabolism and maintain high NO concentrations at the tumor site—a challenge for which no effective means previously existed.
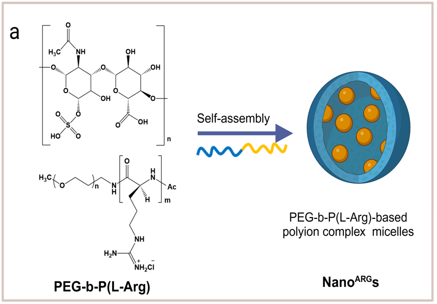
The inventors have successfully addressed this challenge using polyion complex (PIC) micelles with a particle size of approximately 43 nm, formed through the self-assembly of positively charged polyarginine-PEG block copolymers and negatively charged chondroitin sulfate (Fig. a). Following intravenous administration, these PICs selectively accumulate in tumor tissues via the EPR effect. Upon uptake, they release L-arginine via enzymatic reaction, sustaining the generation of high-concentration NO through iNOS metabolism. This process promotes direct killing of cancer cells and facilitates the infiltration of immune cells. Furthermore, when combined with anti-PD-1 antibodies, the technology is expected to exert dramatic anti-tumor synergy by powerfully reprogramming TAMs to the M1 phenotype and transforming the TME from a "Cold" to a "Hot" state.
Data
- Administration of PICs to C26 tumor-bearing mice confirmed that serum NO concentrations increased in proportion to the total administered dose.
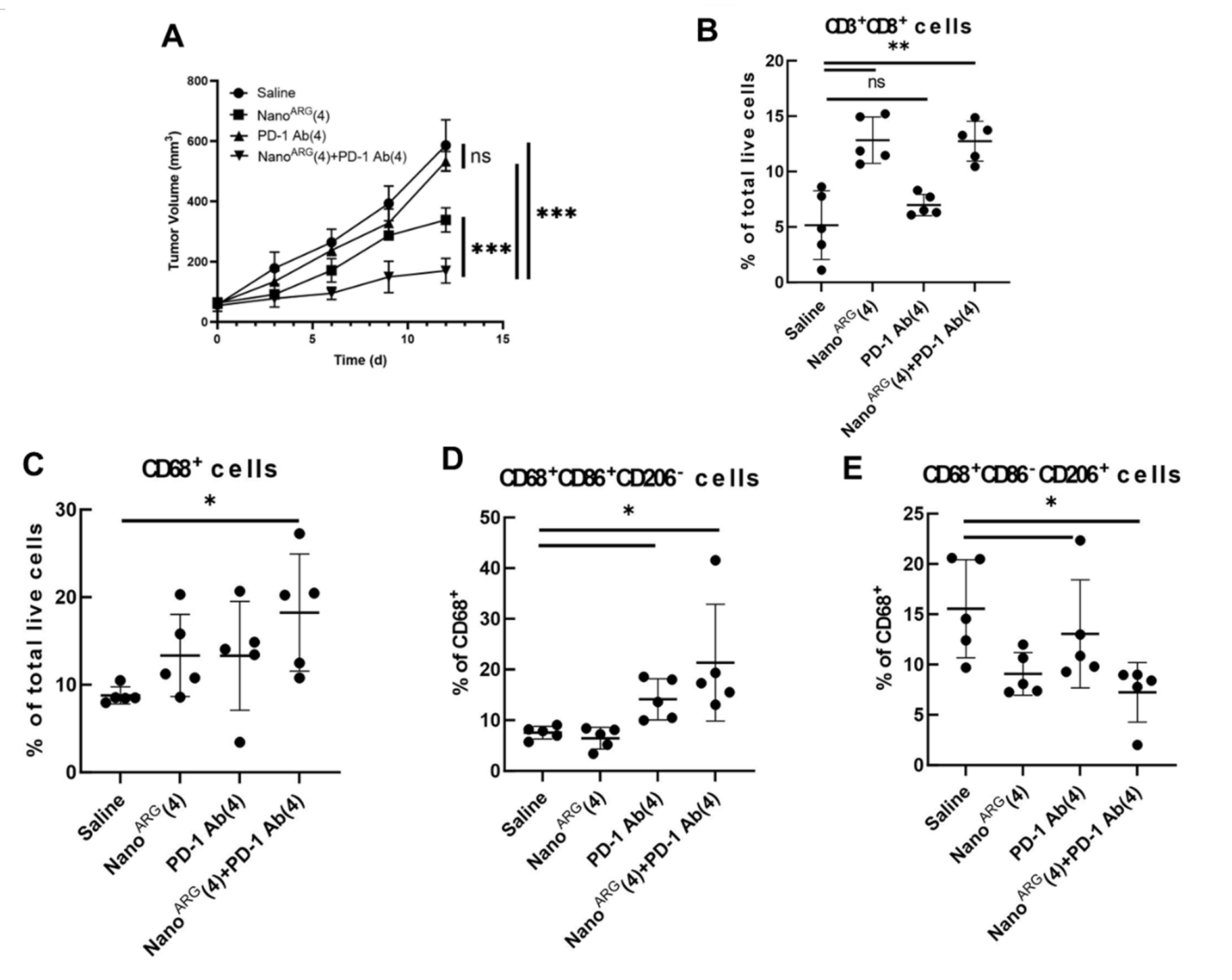
- PIC administration (total dose of 64 mg/kg L-arginine) showed significant tumor growth inhibition, with clear synergy confirmed in combination with anti-PD-1 antibodies (Fig. A).
- Flow Cytometry Analysis: Analysis of single-cell suspensions derived from tumor tissues of treated C26 mice revealed that PICs dramatically increased the infiltration of CD8+ T cells (Fig. B). Moreover, the combination with anti-PD-1 antibodies shifted the macrophage population from an M2-dominant (CD206⁺) phenotype to an activated M1-dominant (CD86⁺) phenotype (Figs. C, D, E).
Expectations
University of Tsukuba is seeking partner companies to commercialize this technology.
- Availability: Evaluation micelles are available under a Material Transfer Agreement (MTA)
- Collaboration Models: Joint Research, Licensing, Technical Advisory.
Meetings with the lead inventor can be arranged upon request.
Principal Investigator
Professor emeritus Yukio Nagasaki: Faculty of Pure and Applied Sciences, University of Tsukuba
Patents and Publications
- Patent: US 10,155,009
- Publication: Ting Mei et al., Science and Technology of Advanced Materials. (2025) 26(1):2538430. [doi]: https://doi.org/10.1080/14686996.2025.2538430